Turning Biomass Into Graphite Could Help Secure US Critical Mineral Supply
National Laboratory of the Rockies Shows How American Refineries Can Use Existing Infrastructure To Boost Domestic Graphite Production

Soft, dark-gray graphite is not just useful in pencils: The highly conductive and heat-resistant mineral is uniquely suited for advanced manufacturing. Yet the United States imports nearly half of the graphite it needs to forge steel, batteries, and manufacturing equipment from China, which currently supplies the majority of global graphite.
But what if the United States could produce that graphite domestically—from waste we would normally toss into a landfill or burn?
That is the goal of new research published in ChemSusChem by the National Laboratory of the Rockies (NLR) and North Carolina State University (NCSU). The research was funded by the U.S. Department of Energy’s Alternative Fuels and Feedstocks Office (formerly BETO).
The research team demonstrated they can use conventional refinery equipment to turn biomass, like forestry and agricultural waste, into high-quality bio-graphite: in other words, a virtually identical match to the imported graphite sought after by battery makers and vehicle manufacturers.
The discovery came about as the research team sought to turn the oil generated from heating biomass into jet fuel. Typically, this oil is difficult to convert to fuel: The reactive compounds within it form a solid coating over the catalysts used to upgrade it. But the research team realized they could target those solid materials and upgrade them into graphite—transforming these “bad actors” into an even more valuable product. And after graphite formation, the process still yields an oil that can be turned into jet fuel and gasoline.
Then, they made a second discovery: The researchers could make the transformation from biomass to graphite occur at a much lower temperature—1,500°C, rather than the traditional 3,000°C—to create significant cost and energy savings.
“What’s novel about this research is that we have shown two viable pathways to produce domestic graphite and fuels as part of the same process,” said Carrie Farberow, an NLR bioeconomy chemical engineer who is a group manager in the laboratory’s catalytic carbon transformation program. “One allows us to turn waste materials into battery-grade graphite, and the other allows for significant energy and cost savings during bio-graphite production.”
With global graphite demand projected to grow 70% by 2028 due to a surge in battery and steel production worldwide, the joint research between NLR and NCSU could help U.S. manufacturers boost production of graphite at an opportune time.
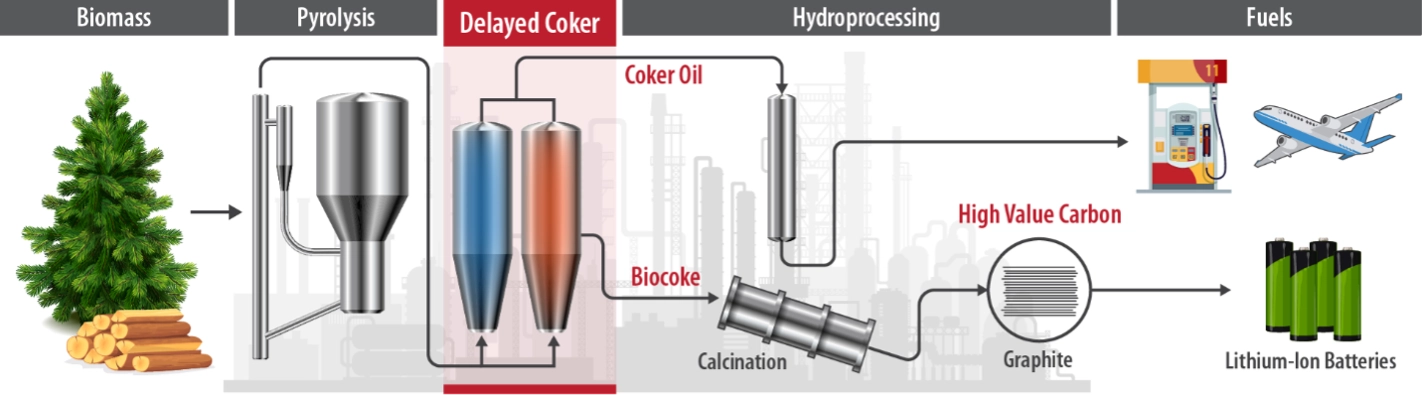
Pathway 1: A Domestic Replacement for Imported Graphite
In the United States, where graphite has not been mined since the 1950s, any domestic graphite production begins with petroleum.
After extracting the valuable components from crude oil, refiners heat the tar-like oil residue to 500°C in a machine called a delayed coker, which squeezes the last bit of fuel from the tar. The byproduct is a solid called coke. The coke is then calcined, or heated at temperatures around 1,000°C, and finally turned into graphite by heating at 3,000°C—nearly 5,500°F. For a sense of scale, that is over half the temperature of the surface of the sun.
Using the same equipment, NLR’s bio-graphite production process shows potential to add to U.S. domestic graphite production using waste biomass, such as forest debris collected to prevent wildfires.
It hinges on a process called fast pyrolysis, where organic materials, such as biomass, are heated to 500°C in an oxygen-free reactor, then quickly cooled to make an oil intermediate. This bio-oil, called “fast pyrolysis oil,” can be upgraded into valuable fuels and chemicals. And it can also form the basis for a drop-in source of American-made bio-graphite, using delayed cokers that refineries have available today.

“During our research, we modeled the delayed coking process for pyrolysis oil derived from biomass,” said Steven Rowland, a former NLR researcher and key contributor to the project. “We saw there is potential for existing petroleum refineries to process the abundant domestic biomass we already collect and upgrade it into high-value carbons like graphite.”
In simple terms, NLR’s bio-graphite production has been tuned to work with traditional refinery equipment. Manufacturers can either use the oil “as is” or blend it with petroleum streams. That can help manufacturers produce more high-quality graphite from their existing feedstocks, adding domestic graphite production across the board.
Pathway 2: Lower-Temperature, Lower-Cost Production
In addition to creating bio-based graphite through a conventional industrial process, NLR and NCSU researchers also developed a second pathway: creating graphite from pyrolysis oil at half the typical temperature using iron as a catalyst. That research is described in a 2025 Energy & Fuels article.
Instead of heating carbon-rich bio-coke to 3,000°C—which requires furnaces themselves to be made of graphite, one of the only materials in the world that can withstand such temperatures—the research team pioneered a method for manufacturers to instead catalytically convert coke to synthetic graphite at temperatures ranging between 1,000°C and 1,500°C.

The lower temperature drastically reduces the energy required for graphite synthesis, typically one of the most expensive aspects of production. It also means manufacturers can use less expensive materials to build and heat the industrial furnaces they use to produce graphite.
While certain steps need to be taken to remove the iron catalyst, such as acid washing, the process can yield substantially more product. Techno-economic analysis published in Bioresource Technology shows it can be financially viable.
A lower-energy, lower-cost path to graphite production could be good news for the American grid. Because graphite is formed from ordered stacks of carbon sheets that can effectively store lithium ions, it is the perfect material for the anode—or negatively charged—side of a lithium-ion battery. These batteries are the leading choice for grid-integrated energy systems, which store energy for later use to ensure our power grid is secure in the face of interruptions or natural disasters.
“The two graphitization pathways we’ve developed have allowed us to demonstrate, from start to finish, how to convert biomass into rechargeable lithium-ion batteries,” said Bertrand Tremolet de Villers, a senior battery chemistry researcher at NLR. “Then, we can characterize and validate those batteries in the lab to make sure they’re safe and high performing.”
“We’re very excited about using these batteries, which we’ve shown can be manufactured with significant cost and energy savings, to support the United States’ electrical grid,” he said.
Turning Waste Into Wealth
Just like catalysts can speed up chemical reactions, U.S. bio-graphite production can get a boost from a surprising source: waste. The scrap wood harvested from forests for wildfire prevention, industrial waste generated by power plants, and even city sewage can turn into graphite to power American battery and steel manufacturing.
Bio-Based Chemicals and Fuels Research

NLR's bio-based chemicals and fuels research supports development of industrially relevant, cost-competitive, and performance-enhanced chemicals and fuels from biomass and waste resources.
“The Department of Energy has reported that the U.S. can produce more than a billion tons of biomass each year in addition to what we currently export. Most of that is amenable to pyrolysis and subsequent graphitization, and it could conservatively yield 100 million tons of battery-grade graphite a year,” Rowland said. “If manufacturers are looking to increase their graphite production, there are a lot of bio-feedstocks that can satisfy demand.”
Research efforts at the laboratory are accelerating accordingly. To scale up bio-graphite production, the laboratory is building a pilot-scale delayed coker that can convert large quantities of pyrolysis oil into graphite and jet fuel. The pilot-scale coker will help show that manufacturers can safely and effectively produce bio-graphite using their existing infrastructure. The graphite it produces will be used in large-format lithium-ion batteries—commonly used for grid-scale energy storage—that will be tested under real-world conditions to prove their effectiveness.
“These processes really have the capability to scale up to meet our future graphite needs,” Farberow said. “With some investment into industrial capabilities, American manufacturing could get the boost it needs.”
Explore our bioenergy and bioeconomy research, and partner with us to develop, translate, and de-risk bioenergy and bioeconomy technologies for commercial adoption.
Last Updated April 28, 2026
