Researchers Describe Protein Structure Microbes Use To Control Light Conversion
Insights on the Protein Complex ‘Photosystem I’ Improve Scientific Understanding of How Cyanobacteria Convert Light Into Energy
Wildfire smoke is teeming with them. Researchers have employed them to develop energy-dense biofuels like rocket, marine, and jet fuels. Scientists have engineered rice paddies that interact differently with them, causing lower methane “burping.” They can be used to extract valuable metals like lithium and copper from plants like seaweed.
What are they?
Microbes. They are everywhere—including in advanced biomanufacturing processes.
Scientists have studied cyanobacteria—a type of microbe—for years as potential candidates for biofuel production, electricity generation, and chemical synthesis. Optimizing cyanobacteria for use in these applications has long been a focus of bioenergy and bioeconomy research at the National Laboratory of the Rockies (NLR), especially how these bacteria capture light and use the energy to drive chemical reactions (known as “photochemistry”).
Recently, researchers from NLR and Montana State University investigated how changes in the structural and chemical properties of cyanobacterial photosystem I (PSI), a protein complex behind that light–energy transformation, influence how it interfaces with sunlight to drive these chemical transformations in the microscopic organisms. Specifically, they observed PSI’s plasticity, which explains how changes in a protein subunit called PsaL affect PSI photochemistry. Cyanobacteria leverage such changes to meet energy demands under shifting environmental conditions.
Their results are published in the article “Modulation of Cyanobacterial Photosystem I Protein Environment and Spectral Capacity in Response to Changes in Electron Flow Pathways and Photon Flux,” with contributors Sharon Smolinski, Monika Tokmina-Lukaszewska, Junia Holland, Zhanjun Guo, Effie Kisgeropoulos, Biran Bothner, Paul King, and Cara Lubner. Funding for this work was provided by the U.S. Department of Energy Office of Basic Energy Sciences.
By better understanding the mechanism of energy control, the study could help address a fundamental knowledge gap and help engineers develop microbes that convert light to energy more efficiently.
“This is important for understanding nature’s strategies for acclimating to different environmental conditions but also applied approaches for converting sunlight into useful products,” said Lubner, a senior NLR chemistry researcher.
Protein Efficiency: How PsaL Affects Photosystem Function
Everywhere we look in nature, changes in one part of a system affect other parts of the system. Beavers, or a lack thereof, affect the ecology of rivers and lakes. As in those ecosystems, cyanobacteria adjust in response to changes in their physical environment and photochemistry. These adjustments affect the mechanisms that turn light into energy.
Natural fluctuations in light quantity and quality affect cyanobacterial behavior. That modulation can lead, under certain conditions, to the production of chemical compounds useful for biomanufacturing applications, including energy production. PSI, a large protein-pigment complex, resides within cyanobacteria to absorb light and convert it into chemical energy.
Science of Biological Energy Conversion Research
NLR's fundamental science of biological energy conversion research capabilities create mechanistic understandings that enable control over biochemical and biophysical pathways and processes in living-organism, cell-free, biohybrid, and catalytic systems.
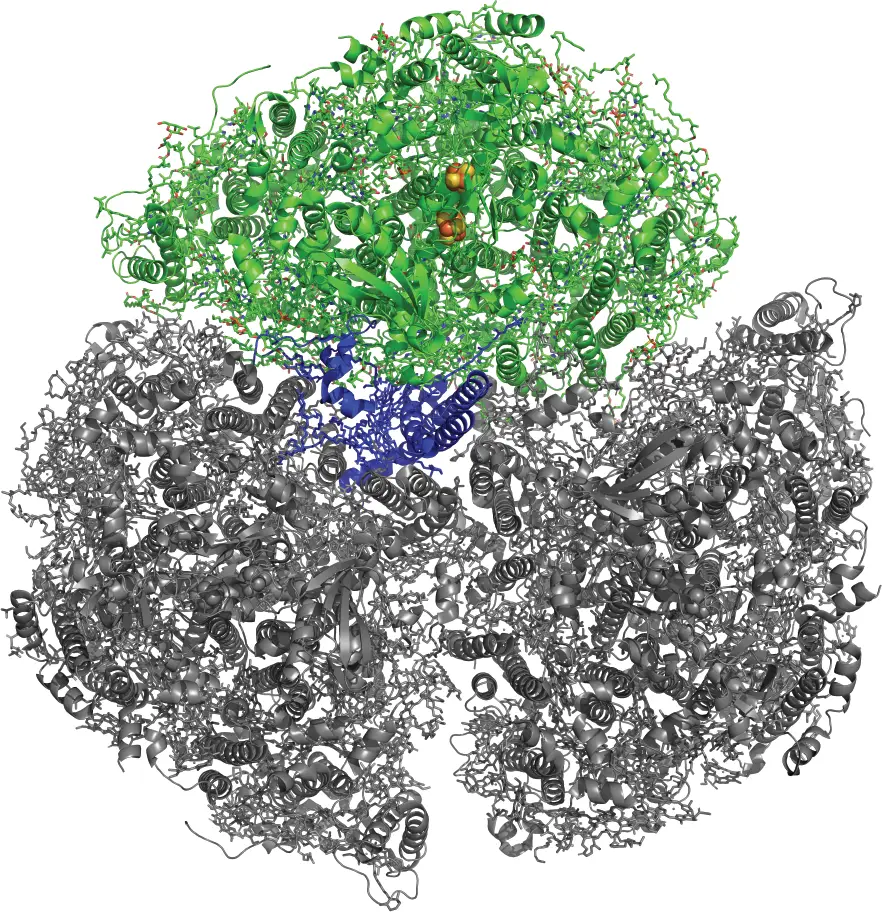
The research team was interested in how PSI responds to high and fluctuating light conditions when cells are missing the oxygen reduction reaction (ORR), a component that allows them to balance energy. As seen in the figure at the beginning of the article, PSI can be monomeric (composed of one protein complex) or trimeric (composed of three protein complexes). The research team investigated these physical properties for clues on photochemistry functionality. Chiefly, they focused on a protein located at the core of the trimer called PsaL that interfaces with the other monomer units. Certain components of PsaL are thought to influence the efficiency of PSI’s photochemical processes. Some scientists aim to tailor cyanobacterial metabolism for energy production, bulk chemicals, pharmaceuticals, and other applications.
To test their hypothesis that the monomerization of PSI is caused by changes to the PsaL environment, the research team subjected monomers and trimers to proteolysis—using an enzyme, pepsin, to break down the proteins into peptides (amino acids). They detected changes in the PsaL pepsin digestion pattern between the wild type and ORR strains, with more peptide fragments present in the ORR PsaL. The team interpreted this to mean the PsaL environment in ORR is easier for the pepsin to access and likely resides further away from the other PSI subunits. The greater accessibility is thought to affect how pigments bind, which impacts the way this PSI interacts with light.
The research team guessed this would happen. They hypothesized that changes to PsaL would affect the physical and photochemical properties of PSI molecules produced from ORR. The changes in PsaL and resulting properties of PSI provide evidence of PSI’s plasticity.
Friends and Neighbors: Changes to PsaL Protein Environments Affect Adjacent Subunits
The research team also observed another key factor in plasticity: how PSI monomers interact with each other through the PsaL subunit.
To identify changes around PsaL that could account for the altered PSI properties, researchers took data from the pepsin digestion experiments and rebuilt PSI-to-PSI interactions. They noted changes in how monomers interact with each other. PSI’s light-harvesting “antenna” is spread across the entire protein complex, and researchers know that PsaL’s environment is altered, signaling implications for how energy is transferred through discrete points within the complex.
In other words, the team discovered that even subtle changes to PsaL in cyanobacteria affect PSI structure and function.
Carotenoids: Essential Pigments Are Abundant in PSI Molecules
Carotenoids are essential pigments in photosynthetic organisms like cyanobacteria that protect against cell damage from light and absorb light for photosynthesis.
“I think of carotenoids as nature’s sunscreen,” Lubner said. “They help shield the sensitive machinery inside the cell from too much light.”
The researchers measured carotenoid abundance in the PSI in samples via their absorbance at 400 to 500 nanometers, the region for carotenoid absorption. Carotenoids showed up in increased numbers in some ORR PSI molecules under fluctuating (more intense) light conditions and, in moderate light conditions, to a smaller extent versus the wild type. When the team analyzed the makeup of the carotenoids, they noticed changes in the types of carotenoids present, reflecting differences from the overall ORR PSI molecules. Light conditions and loss of the ORR affected carotenoid abundance, which in turn altered PSI light absorption properties.
“The ORR strain is lacking a pathway to deal with excess light, so it seemed that the ORR cells found another way to compensate by increasing the amount of carotenoid sunscreen,” Lubner explained. “Changing the compositions of carotenoids restructured the cells and PSI to use specific wavelengths of light.”
Plasticity in PSI Molecules
These findings all show that PSI exhibits plasticity. Changes to PsaL affect the physical and photochemical properties of PSI molecules. PsaL is more exposed, and more available, in the ORR strain compared to the wild type, which can affect the orientation in the subunit, among other implications. The availability of PsaL, the effect of that availability on adjacent components, and the abundance of carotenoids all showcase how microbes can adjust PSI structure. These small changes all work to ensure the energetic demands of cyanobacteria are met under fluctuating energetic conditions.
“For a photosynthetic microbe to generate useful products, it needs to transform the light coming in into other forms of energy,” Lubner said. “PSI is where this conversion happens, but PSI can send that transformed energy to different pathways. There are several alternative pathways that on the surface appear to be ‘wasteful,’ because instead of using the energy for making reduced products, they use it to protect the cell and machinery against specific conditions like excess light. (This is what ORR does.) Our work shows what happens when these pathways are removed and helps inform better strategies for converting sunlight into chemicals.”
Discover other NLR science of biological energy conversion research, including on pathways of electron transfer. Also, learn more about Basic Energy Sciences at NLR and about the U.S. Department of Energy's Office of Science Basic Energy Sciences program. Read “Modulation of Cyanobacterial Photosystem I Protein Environment and Spectral Capacity in Response to Changes in Electron Flow Pathways and Photon Flux” in the Journal of Biological Chemistry.
Last Updated April 28, 2026

