New ‘Atlas’ Will Catalog Proteins That Bind to Rare Earth Elements
Bioprospecting Tool Could Boost Critical Mineral Refining

Throughout the 1800s, naturalists journeyed to far-flung corners of the world to catalog birds, mammals, minerals, and plants. Now, a team of U.S. researchers is assembling a catalog of their own: an atlas of naturally occurring proteins capable of binding to rare earth elements.
Researchers are aiming to find proteins that bind with the right strength to rare earth elements. Once these proteins are identified, they could be incorporated into bioseparations systems that can extract desirable metals from domestically sourced wastes.
Led by the National Laboratory of the Rockies (NLR) and supported by Pacific Northwest National Laboratory (PNNL), the creation of the first-ever Microbial Rare Earth Element Atlas could help address the list of 60 critical minerals identified by the United States Geological Survey as vital to the national economy but at risk of disrupted supply chains.
Funded with $2 million from the U.S. Department of Energy’s Office of Science, the atlas may help bolster the nation’s supply of neodymium, praseodymium, dysprosium, terbium, and other members of the lanthanide class of elements. These elements are essential to creating the powerful magnets used in electrical generators, batteries, computers, and MRI scanners—but the United States largely relies on importing these elements from China.
Using machine learning, the NLR/PNNL team is developing a “bioprospecting” tool that can identify geographic areas within the United States likely to be rich in these naturally occurring proteins. Then, they will develop methods to optimize the metal-binding process, with a goal of using specific proteins to bioaccumulate metals from domestic wastes.
The work, part of NLR’s critical minerals research focus, aims to enable rare earth mineral accumulation and refining right here in the United States: a step toward an affordable, fully domestic supply of critical minerals.
Step One: Building a Microbial Map
Tucked within the complex structure of proteins are sites that can bind to specific metals, like the rare earth elements that power batteries, homes, vehicles, and the grid. Recently, researchers have discovered that the proteins within microbes, the most abundant life form on Earth, recognize and use these rare earth elements.
In other words, the microbes found within our soil represent untapped potential to find and bind to critical minerals—if we can find them.
“In the first step of the project, we are truly building a map, a geographic and a phylogenetic map, of the microbes and proteins that utilize rare earth elements,” said Alli Werner, a senior biological engineer leading NLR’s work on the project.
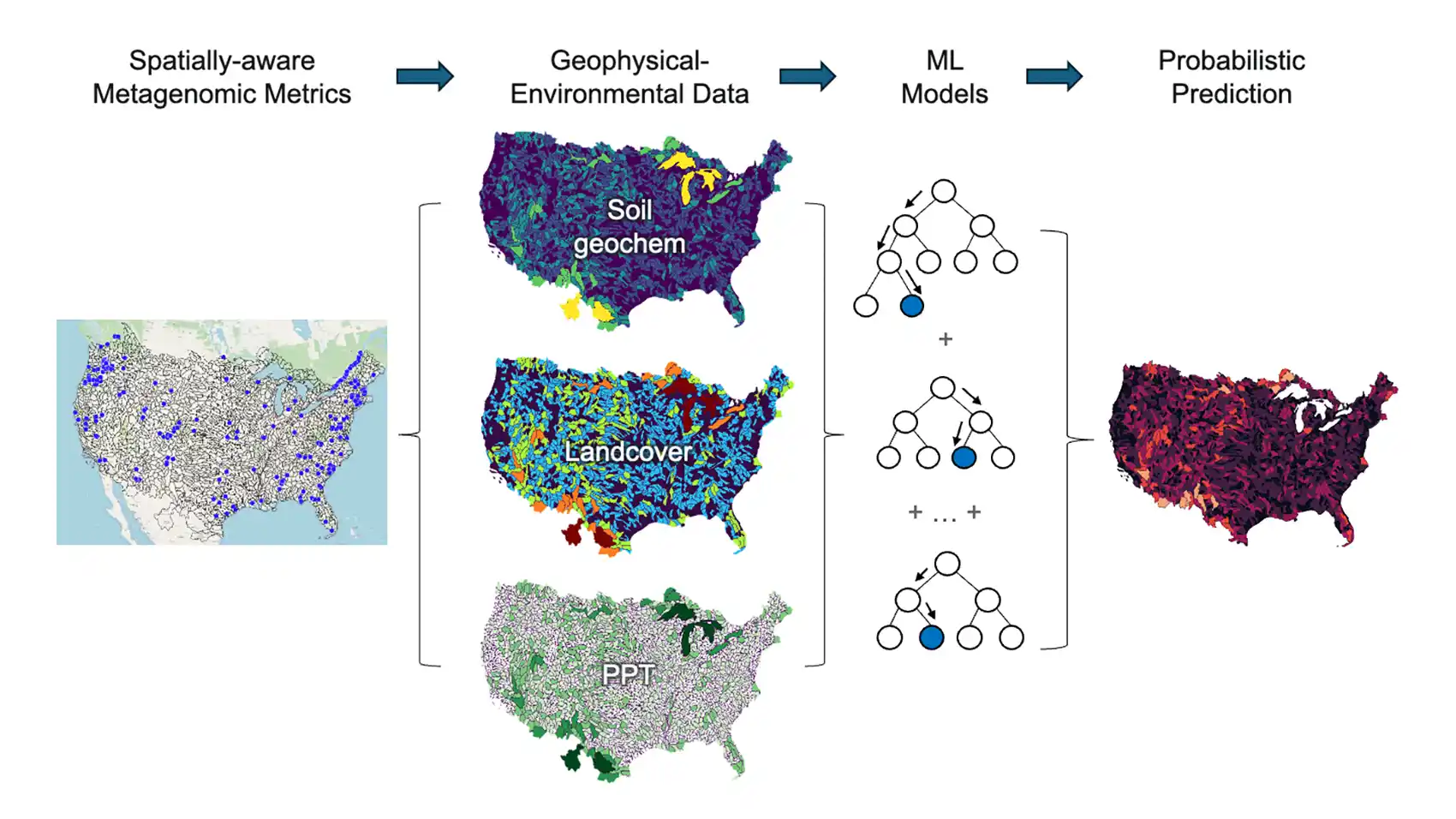
“Scientists only recently discovered that some proteins bind specifically to rare earth elements,” she continued. “We want to understand the natural diversity of these proteins so that we can predict where to find more and train models to redesign them for better metal-refining processes.”
Starting with a map of the nation, the team will leverage PNNL’s large environmental datasets to layer on metagenomic, geophysical, and environmental data. Combined, these layers will allow researchers to understand whether soil chemistry is correlated with abundant proteins with the correct structures to bind to rare earths.
Then, they will feed the data into a machine-learning-enabled model to create a “precision bioprospecting” tool capable of predicting where researchers should dig to find a specific metal-binding protein.
Step Two: Identifying Promising Metal-Binding Proteins
With environmental samples in hand, researchers can sequence the microbial DNA and pull out potential new metal-binding proteins. This is where NLR’s high-throughput, robotics-enabled protein validation pipeline comes into play.
“Let's say we sample some dirt and we find a sequence that looks like it encodes a protein that binds to rare earths,” Werner said. “That sequence then forms one in a growing library of candidates. But to make the library useful, we have to then validate the genetic sequence by asking: Does that protein bind to rare earths? How tightly does it bind? Does it bind selectively, or will it bind to many different elements? And how many times can it bind and release before degrading?”
Using robotic-assisted experimental pipelines, NLR researchers can rapidly screen hundreds of protein–metal combinations to identify the affinity and selectivity of the bonds they form with metals, as well as the stability of the protein after binding and releasing.

Researchers are aiming to find proteins that bind with just the right strength to just the metals they seek. And they are most interested in proteins that remain stable over multiple cycles of absorbing and releasing rare earth elements, which will be most useful in rare earth element refining.
Once these proteins are identified, they could be incorporated into bioseparations systems—like one being designed at NLR—that can extract desirable metals from domestically sourced wastes like old electronics, mine tailings, and industrial byproducts.
Step Three: Tackling Real-World Refining Challenges
As a final step, researchers will aim to understand if the metal-binding proteins they find play an important role in the biology of the microbes in which they were found. This information will help them determine how to bioaccumulate desirable metals right here in the United States.
“Just because a protein binds to a metal doesn’t mean it’s doing something functional,” Werner explained. “We want to understand how the microbe is using the metals and proteins in concert so we can engineer the biosystem as a whole.”

For example, researchers could use rare earth-binding proteins to concentrate the metals found in dilute resources, such as mine tailings or wastewater. But researchers need to understand the fundamental ways in which proteins bind with metals, so they can control and optimize the process.
Once they crack this question, Werner explained, it could be possible to reduce the cost and resources required to produce rare earths domestically—including from waste we already produce.
“Refining rare earth elements today is expensive and waste-intensive,” Werner said. “For example, it requires a huge amount of chemical solvent to slowly separate similar metals from each other. Proteins that selectively bind to the rare earth elements could help us perform the same separation in far fewer steps and with a much lower chemical demand.”
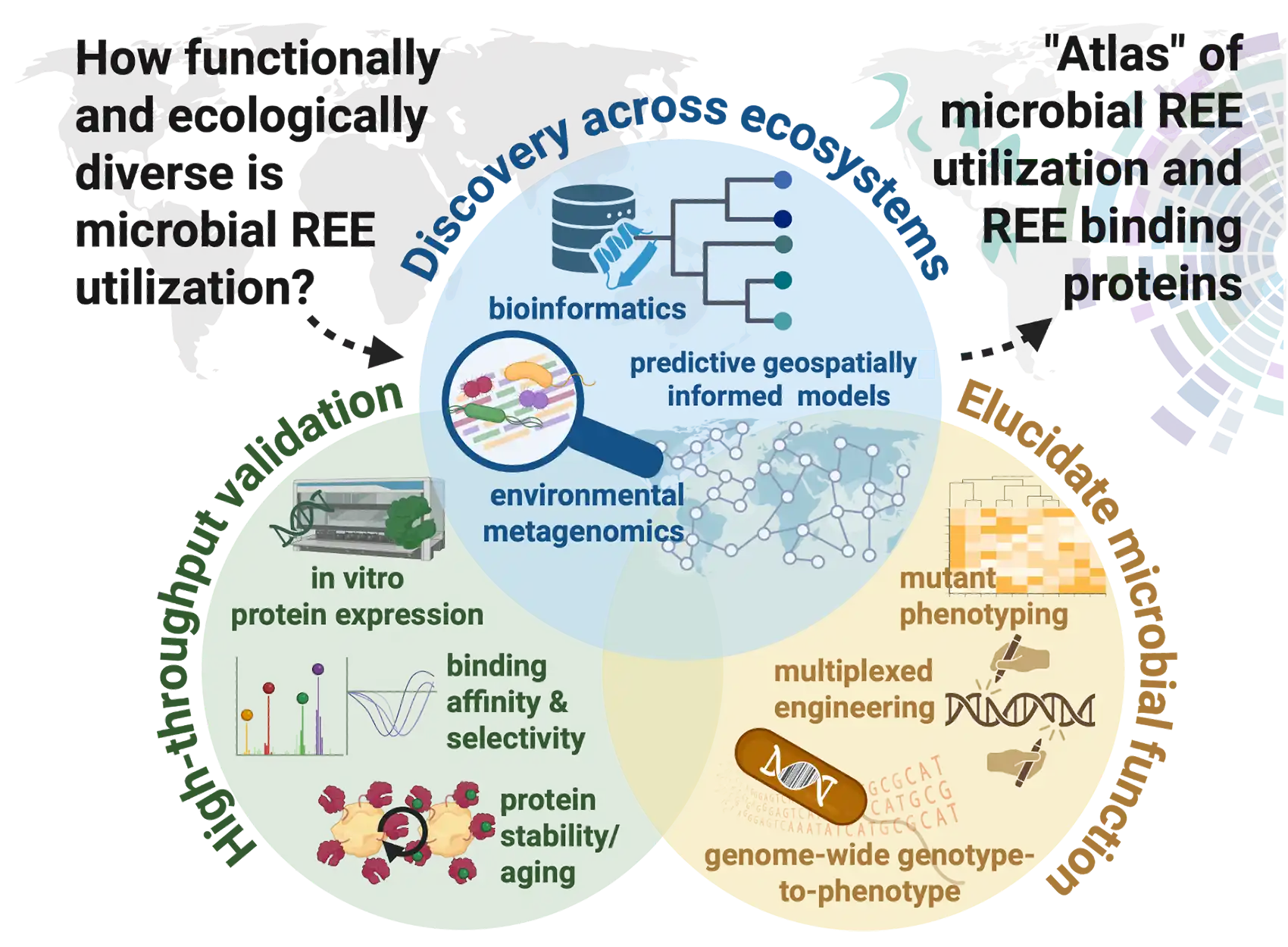
With a lower-cost refining process in place, industry partners could next begin separating rare earth elements from domestic waste that is rich in them, such as coal fly ash and mine tailings.
“If we could tap into those waste streams, then we could replace much of what we currently import from China with our own domestic supply,” Werner said.
Colleagues agree the research shows promise to boost the nation’s domestic critical mineral supply.
“It’s extremely exciting to have the opportunity to integrate synthetic biology with continental-scale, AI-enabled ecosystem science,” said James Stegen, a senior PNNL scientist on the research team. “I feel this partnership could transform our capacity to leverage the nation as a reservoir of biological novelty and create a vital increase in domestic supplies of critical minerals.”
Discover other biomanufacturing innovations for mining, industry, and agriculture, as well as NLR’s biological energy conversion and critical minerals research. Partner with NLR to develop, translate, and de-risk bioenergy and biomanufacturing technologies for commercial adoption.
Last Updated April 28, 2026
