Why Use Living Cells? NLR Is Making Chemicals With Enzymes Alone
Researchers Address Technical Challenges for ‘Cell-Free’ Biomanufacturing—Which Could Reduce Costs of Bio-Based Chemicals

Today’s nearly $70 billion U.S. biofuels economy is powered by two technology toolboxes.
Biochemical technologies—used to produce around 17 billion gallons of ethanol annually—leverage microorganisms to convert plant biomass sugars into alcohols, other biofuels, or chemicals. Chemical technologies, the second toolbox, use catalysts to turn biomass and wastes into similar target products.
The implements in these toolboxes are powerful, and they continue to expand. Since its biomass program was founded in the 1980s, the U.S. Department of Energy’s National Laboratory of the Rockies (NLR) has conducted research contributing to numerous new capabilities, products, and efficiencies. Just since 2020, NLR has been issued over 294 biotechnology patents, which industries are licensing for their growth in the open market.
And for the past five years, NLR has been conducting fundamental research and development toward a third technology toolbox. “Cell-free” biomanufacturing, as it is often called, could open new doors for turning U.S. biomass and waste into usable chemicals.
At a high level, cell-free biomanufacturing means using enzymes (biological catalysts) to drive the conversion of biomass-derived feedstocks without cells. Enzymes liberated from the constraints of living organisms can provide a significant advantage because they are less sensitive to cytotoxicity of products and feedstocks, product transport, complex metabolism, and competing pathways, among others.
In short, there are no cells, no organelles, and no DNA. Instead, a handpicked lineup of enzymes drives controlled and extremely precise reaction chains. Together, they turn plant biomass-derived sugars and compounds into high yields of specific chemicals—including some impossible or costly to make with today’s technologies.
“Cell-free biocatalysis for biomanufacturing—or cell-free biomanufacturing—essentially operates enzymatic cascades without the constraints of a living cell. This is a technology that could transform the bioenergy sector but also pharma and fine chemicals,” explained Yannick Bomble, an NLR principal scientist and an R&D leader in cell-free biomanufacturing.
Indeed, cell-free biomanufacturing could unlock dramatic increases in efficiency through highly specific and fast biochemistry. But moving cell-free concepts out of the laboratory demands rigorous science to clear technical hurdles.
To overcome these hurdles, NLR bioeconomy scientists are pioneering new cell-free approaches and technologies using molecular engineering, machine learning, and high-throughput techniques.

What Is the Cell-Free Opportunity? Precise Reaction Cascades Outside of Living Cells
In NLR’s Field Test Laboratory Building, NLR researcher Hunter Harrington operates liquid-handling robots that pipette enzyme samples into plates. Working together, the robots can prepare samples of over 1,000 enzyme variants in less than an hour—much faster than a human could. The goal is to quickly sort and then characterize as many enzyme samples as possible.
“We are looking for changes in the metabolites,” explained colleague Sam Mallinson. “Some molecules absorb at a specific wavelength, and then they undergo the reaction with a detectable signal change, which is what we measure.”
Capturing such data points—several thousand in a single day—enables the team to spend their time building and then analyzing rich datasets on enzyme properties. Using artificial intelligence, they screen the resulting libraries for interesting enzyme traits, highlighting promising designs and functions in a matter of minutes. What makes these enzymes work so well in nature? What design features make them tough? And how might those abilities be improved in an even more extreme environment—removed from a parent cell entirely and potentially exposed to solvents?

“The idea is to narrow down promising enzyme candidates to a small enough number, which we can then use to do manual testing,” Mallinson added. “Then we test them in reaction pathways.”
Enzymes are a key component of life. They are proteins that are folded, twisted, stretched, and crumpled into complex 3D shapes that facilitate specific chemical reactions. Like a wrench tightens a bolt or a saw cuts lumber, aspects of the molecular structure of each enzyme determine the reaction it triggers. Together, millions of enzymes produced by cells in our bodies use that functionality to complete the many chemical tasks needed to regulate blood sugar, digest food, or heal tissues.
The cell-free systems that NLR is developing are not so different from the enzyme-driven reactions in our bodies, except they will never be used in a cell.
Instead, a dozen or so enzymes—each with a specific purpose and often engineered to function best cell-free (i.e., “exozymes”)— are dumped into a bioreactor. The researchers mix in sugars from corn stover, wood chips, or other plant materials, along with some energy carriers, water, or solvent. Once submerged—and in direct contact with the sugars—the enzymes start their work. One binds to the sugars to release an intermediate. Another turns an acid to an aldehyde. Soon, a customized cascade of reactions unfolds—one with limited inhibitions—with each enzyme completing a specific task on the path from sugar to final product.
In effect, NLR researchers have eliminated background metabolic processes of microbes completely. The researchers control every step of the reaction—making just what they want, right when they want it.
The challenge, according to Bomble, is improving enzyme systems to sustain those yields—on cue—and at a price point attractive to industry. In other words, what molecular folds and twists make enzymes both stable and efficient? How can they be easily manufactured? How can industry be sure they last?
Here are four key hurdles NLR is working to overcome to unlock the potential of cell-free biomanufacturing.
Hurdle #1: Designing Stable, High-Performing Enzyme Systems
NLR is discovering enzymes with traits best adapted to cell-free systems—and developing methods to make them synthetically.
To meet industry’s high standards, the various enzymes in a cell-free system cannot easily break down over time or when exposed to solvent. They must perform for days so companies can recoup the cost to make them. Every enzyme in the system must be tunable so industry can engineer highly precise reactions.

“Let’s say there’s a chemical reaction we want to catalyze. We likely already know of enzymes needed to make it happen, or those that have this activity, but the yields are pretty poor,” Bomble said. “Can we engineer an enzyme to increase the yield by enhancing its catalytic efficiency? We most likely can, but we need to be able to screen tens of thousands of enzyme variants or more, which is where our robotics and high-throughput capabilities come in.”
Using the high-throughput techniques and machine learning described earlier, the team is screening tens of thousands of variants to determine the best combinations of robust enzymes to catalyze a series of chemical reactions. For example, in a recent article in collaboration with industrial partner eXoZymes Inc., they showed that thermophilic enzymes, owing to their greater stability, operated six times longer than those from bacteria from milder environments, nearly doubling terpene yields. Plus, the enzymes exhibited much higher solvent tolerance.
Yields that high for these types of compounds are nearly unheard of using a microbial chassis, Bomble said, illustrating the potential power of cell-free biomanufacturing.
According to him, thermophilic enzymes, naturally stable at high temperatures, offer blueprints for further improving the enzymes—applicable to making other products to serve other industries.
“We’re hoping that we can learn the design principles,” he explained. “For example, if we found interesting patterns in an enzyme from a hot spring that's super stable, we could just make similar mutations to other enzymes we are designing for our cell free systems.”
Hurdle #2: Engineering Cheap, Broadly Effective Synthetic Cofactors
NLR is creating a library of cofactors with key traits: stability, solubility, affordability, and compatibility (redox potentials).
Cell-free systems require more than stable and tunable enzymes. These systems need cofactors too, the nonprotein “helper molecules” that trigger some enzymes’ activity. Even though cofactors are continuously recycled in these pathways, priming the system with natural cofactors can be costly for commodity chemicals. So, the researchers are identifying cofactor replacements that are compatible with their enzymes.
Aside from better enzymes, cofactors are probably the biggest cost driver in productive cell-free systems for commodity chemicals, Bomble said.
“Cofactors need to be cheap and stable. We need to engineer them for solubility for separation. We can also fine-tune their redox potential,” Bomble said. “Those are intrinsic traits we want cofactors to have. Some reactions just aren’t possible using natural cofactors, so we are synthesizing our own to force reactions to take certain pathways.”
NLR, with partners at Colorado State University, has been turning to artificial intelligence and advanced models to help make cofactor design predictions. What cofactors work with the enzymes NLR is designing? Do they have the traits needed for industry success?
In a 2025 Green Chemistry article, the team used density functional theory and computational modeling to identify 132 nicotinamide cofactors that can be made synthetically. Since the paper was published, that library of cofactor candidates has been expanded to more than 1,000.
“Our goal is to make it easy to predict the cofactor properties before researchers have to think about synthesizing them,” Bomble said. “That could go a long way in streamlining the process to make cofactors more affordable and viable at industrial scales.”

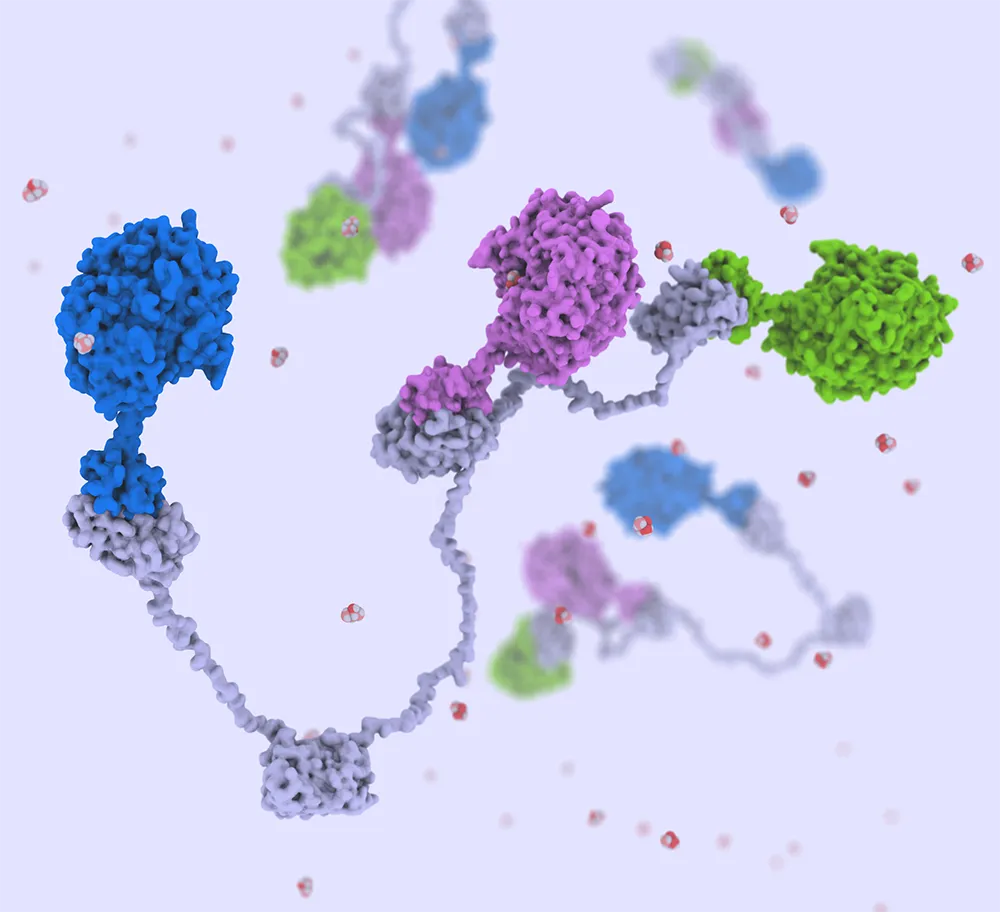
Hurdle #3: Identifying a Cheap—and Universal—Enzyme Immobilization Technique
NLR is developing a flexible technique—inspired by thermophilic bacteria—to attach dozens of different enzymes to a single bead.
Using cell-free techniques to achieve high yields of specific chemicals can be far harder than just dumping sugars into a tank, adding a dozen enzymes, mixing, and expecting the system to work efficiently. Doing that could destroy some of the enzymes in a couple of hours—not nearly long enough for a company to return a profit. It can also make product recovery more difficult, as enzymes are free floating in the reaction vessel.
Preferably, those enzymes need to be attached to something, held in place, and immobilized. This extends their operating lifetimes, makes them stable, and enables engineers to recover the products while retaining enzymes in the reactor. Those techniques themselves represent highly complex scientific challenges—challenges the NLR team is taking great strides to solve.
“What we're hoping to do with our tethering technique is to have an immobilization approach that's cheap and broadly applicable to all of the enzymes we use,” Bomble explained. “For us, this addresses one of the biggest barriers to making this kind of system work: the lack of a universally applicable immobilization approach. A single technique can be used to immobilize one or two enzymes but doing so successfully for 15-plus enzymes with the same technique is really hard if not impossible. We are hoping our novel technology can do it.”
In essence, the team’s approach involves specifically attaching dozens of enzymes to flexible protein “strings” that can bind microscopic beads—which companies can modify to tailor cell-free systems to their unique business cases. The model is cellulosomes, interesting structures naturally found in thermophilic bacteria wherein enzymes are tied to “dockerins” designed to bind to a scaffold using a “cohesin.” An NLR patent illustrates the broad applicability of this scaffold technology, and a forthcoming Bioresource Technology article describes its application to make 2,3-butanediol.
“The strength of our approach is that we can do this with no additional cost except for the cost of making that small extra protein,” he explained. “And it lets us make products at the same levels that we can with untethered systems—but with that added stability and the ability to reuse proteins.”
Hurdle #4: Combining the Best of Biology and Chemistry
To push efficiency even higher, NLR is engineering systems that use both enzymes and inorganic catalysts.
Without a doubt, cell-free biomanufacturing could be powerful alone. But NLR scientists also imagine a future where chemistry and biology can be wielded together to make chemicals and other bioproducts.
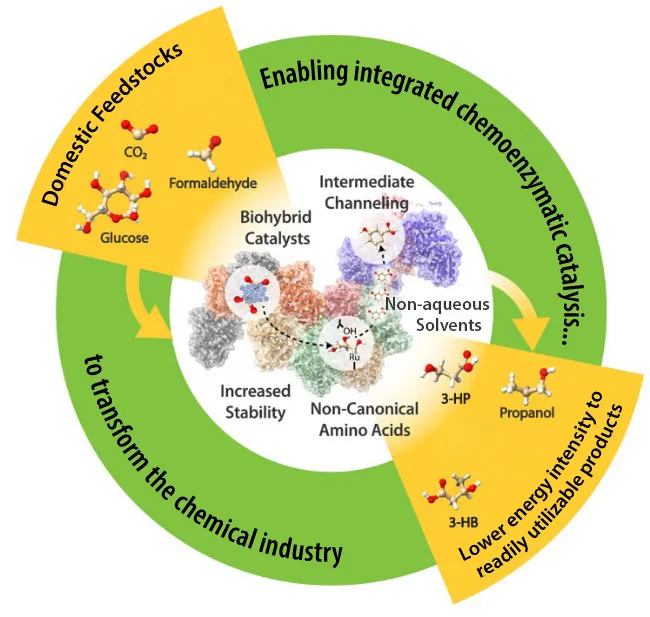
“What is powerful about cell-free biomanufacturing is that you can actually combine chemical catalysis and enzymes in the same pot, because they are much more compatible,” he said, “essentially mixing the two together to leverage the best of both worlds.”
Bomble and collaborators are exploring this idea as part of NLR’s Laboratory Directed Research and Development program, which is designed to accelerate R&D on potentially disruptive technologies.
Where enzymes enable highly specific reactions, chemical technologies (remember that other biomanufacturing toolbox?) let engineers pump in energy, speeding up specific steps in a reaction chain.
“We think that in some cases we can achieve much higher yields with chemoenzymatic approaches than would be possible with biology or chemistry alone,” Bomble said. “An inefficient 12-step chemicals synthesis route with tons of carbon wasted could potentially be replaced by a handful of cell-free, enzymatic steps and a single chemical step—a much shorter route but most importantly a much more carbon-efficient one.”
Although research is ongoing, the team is attuned to how it could potentially benefit a range of industries. For example, it could enable biochemical and pharmaceutical companies to replace some redox enzymes with chemical catalysts to avoid the need for cofactors altogether. It could help a chemical company turn an intermediate from chemical catalysis into next-generation plastics by adding a final enzyme step—potentially very difficult with chemistry or cell-free pathways alone.
“You could do nearly everything with a cell-free enzyme system alone, but there are a lot of transformations that will not be optimal,” Bomble explained. “By combining those two, we aim to harness the advantages of both.”
Going Cell-Free With Industry: NLR Partnerships Prepare Cell-Free Toolbox for Commercial Impact
Bomble sees value in growing the circle of partnerships to accelerate R&D, enhance industry processes, and develop new technologies.
“We already work on this with the University of Colorado, the Colorado School of Mines, Colorado State University, and Lawrence Livermore National Laboratory,” Bomble explained. “So, we’re kind of building a team to address the most urgent hurdles. We also already have a very fruitful partnership with eXoZymes Inc., one of the leaders in this field, and we need others to help.”
Through the Shell GameChanger Accelerator Powered by NLR, researchers worked with eXoZymes, previously Invizyne, to help improve isobutanol separation in a cell-free system. NLR also worked with eXoZymes on a U.S. Department of Energy Technology Commercialization Fund project to demonstrate that protein production and storage at scale for cell-free biomanufacturing is possible at a reasonable cost.
“Cell-free biomanufacturing has the potential to expand how fuels, chemicals, and materials are produced by enabling greater control, speed, and process flexibility,” explained Tyler Korman, the chief science officer and one of the founders of eXoZymes Inc. “NLR’s experience in biocatalysis, together with its expertise in process design and modeling, not only supports our efforts to advance cell-free programs and evaluate opportunities to improve process performance and economics relative to conventional biological approaches but helps push the field further. The collaboration between eXoZymes Inc. and NLR resulted in a meaningful step forward for biomanufacturing innovation, and it has been a pleasure working with Yannick and his excellent team.”
Building a third biomanufacturing toolbox will not happen in a silo. Commercial development will require rigorous piloting and scale-up, not to mention industry collaboration to help address the most pressing technical challenges as they explore potential applications.
With the attention of specialized scientists, amplified by the strengths and capabilities of research institutions, it has potential to make a real difference.
Learn how to partner with NLR bioeconomy researchers to develop, translate, and de-risk cell-free innovations for market readiness.
The research described in this feature was supported by the U.S. Department of Energy Alternative Fuels and Feedstocks Office, an Energy Technology Commercialization Fund project, NLR’s Laboratory Directed Research and Development Program, and a Shell GameChanger Accelerator project. Learn more about NLR’s Science of Biological Energy Conversion Research, including biosystems design.
Last Updated April 28, 2026
